Welcome to MSU Patient-Centered Research
We believe in the patient-centered research approach – putting the patient at the center of any collaboration with industry, academia, and government agencies to improve the quality of life of ALL affected by myositis. Learn more about the research we are involved in, the impact it can have, and how you can get involved.

MSU Research Philosophy
- Patients are the experts in their disease and their data and experiences drive research
- We keep the patient front and center in every collaboration with industry, corporate partners, academia, and other organizations
- Where appropriate, patients will be fairly compensated for sharing their experiences
We will be intentional in our partnerships and research funding:
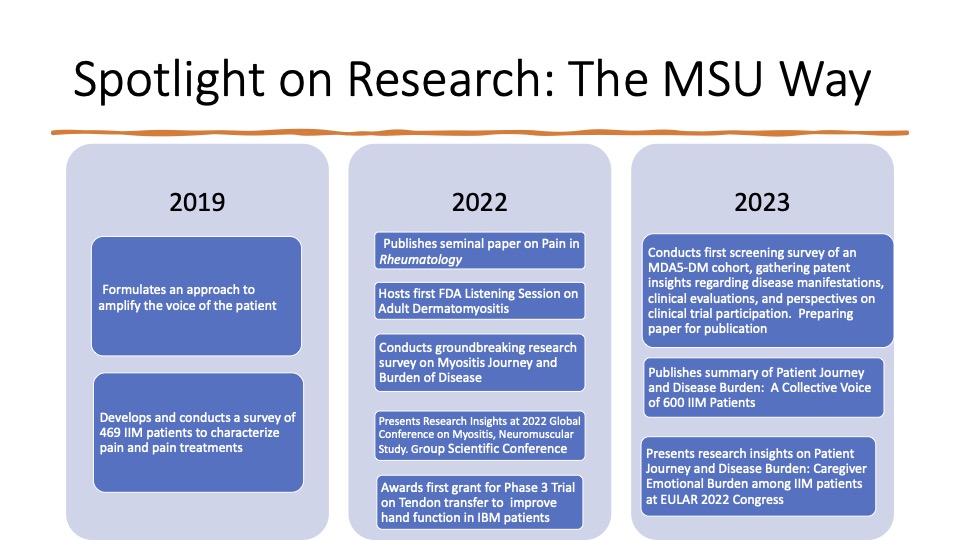
- Amplifies the voice of our diverse patient population
- Enables MSU to influence the direction of research efforts
- Provides a clear line of sight to impact on the myositis community
MSU Research Approach
MSU will center research efforts on the everyday improvement of patient quality of life (QoL)
Prioritizing patient-centered research initiatives that would benefit the quality of life of the myositis patient, among those:
- Crowdsourcing of patients needs/wants by myositis type to capture the voice of the patient (VoP)
- Burden of care research that addresses gaps in myositis care management
Providing research grants through MSU or affiliate partners for studies that impact near term functional diagnostic and treatment improvements
- Novel clinical initiatives to prolong function in myositis patients
- International MSA standardization for diagnosis & treatment of myositis
Partnering with academia, industry, and government agencies to understand disease evolution and subtype response to advance research in drug treatment and improved clinical trial design
- Collaboration with AllStripes on their DM and IBM research programs
- FDA listening session on Adult Dermatomyositis

Patient-Led
Research
The MSU Research Team
- Lynn Wilson: President, Director of Patient-Centered Research
- Manuel Lubinus: PhD, Chief Science Officer
- Izandra Rudolph-Heard: M Ed, Community Outreach, Mental Health
- Pai Hu: Data & Marketing Strategy
- Stephen Moore: PhD, Neuroscientist UAB: CME, DM Projects
- Benita Moyers: Vice-President and Secretary, Anti-MDA5 DM Project Manager
Medical Advisors
- Salman Bhai, MD, UT Southwestern, Neurology
- Abhiram Bhashyam, MD, PhD, Mass General, Hand and Upper Extremity Surgeon
- Lauren Graham: MD, PhD, UAB Dermatology
- Shobhana Natarajan, PhD, Genetics, Sr. Medical Affairs Director, Reata
Your Data & Experiences Drive Research
For more information or to contact the MSU Research Team, email Research@Understandingmyositis.org.
Stay tuned for important updates including new survey results and more.